A double-headed arrow between Lewis structures indicates that they are resonance forms. The actual electronic structure of the molecule (the average of the resonance forms) is called a resonance hybrid of the individual resonance forms. The individual Lewis structures are called resonance structures. Instead, the concept of resonance is used: The actual distribution of electrons in each of the nitrogen–oxygen bonds in NO 2 − is the average of a double bond and a single bond. It is not possible to write a single Lewis structure for NO 2 − in which nitrogen has an octet, and both bonds are equivalent. However, experiments show that both N–O bonds in NO 2 − have the same strength and length, and are identical in all other properties. A double bond between two atoms is shorter (and stronger) than a single bond between the same two atoms. If nitrite ions contain a single and a double bond, the two bond lengths are expected to be different. The Lewis structure of a nitrite anion (NO 2 −) may actually be drawn in two different ways, distinguished by the locations of the N–O and N=O bonds. 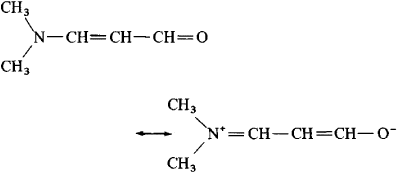
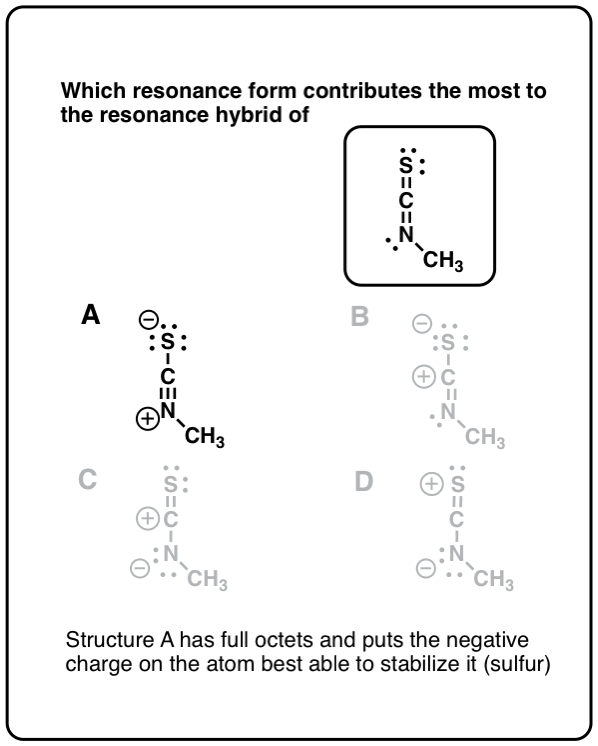
Resonance Structures and Resonance Hybrids Thirdly, structures that minimize formal charges are more stable.įinally, structures carrying negative charges on more electronegative atoms and positive charges on less electronegative atoms are more stable.Īccording to the theory of resonance, if two or more Lewis structures with the same arrangement of atoms can be written for a molecule, ion, or radical, the actual distribution of electrons is an average of that shown by the various Lewis structures. Secondly, structures with a greater number of covalent bonds are more stable. Certain preferences are used to estimate the relative energies of various contributing structures.įirstly, structures in which all atoms have filled valence shells are more stable. If a contributing structure is lower in energy than another, it more closely resembles the actual molecular structure. Such delocalization results in resonance stabilization-that is, a molecule with a lower potential energy than that of any theoretical non-delocalized structure. Resonance structures are identified by ‘electron pushing’, or transforming lone pairs into bonds and vice versa, as denoted with curved arrows. In these molecules, the actual structure is the weighted average or a hybrid of its resonance structures. These double-headed arrows can be thought of as commas and should not be confused with resonance structures being in equilibrium. The spatial positions of all of the component atoms remain the same however, the valence electrons are distributed differently. These multiple Lewis structures of sulfur trioxide are called resonance structures or contributing structures. However, to reach a full octet for the sulfur, an additional bond must be formed between sulfur and one of the oxygen atoms. The single bonds between each oxygen and the central sulfur atoms satisfy the octet for the oxygen atoms. These compounds can be represented accurately with multiple Lewis structures.Ĭonsider the Lewis structure for sulfur trioxide. However, in particular compounds, some electrons are delocalized over multiple bonds or atoms rather than localized to a specific bond or atom. 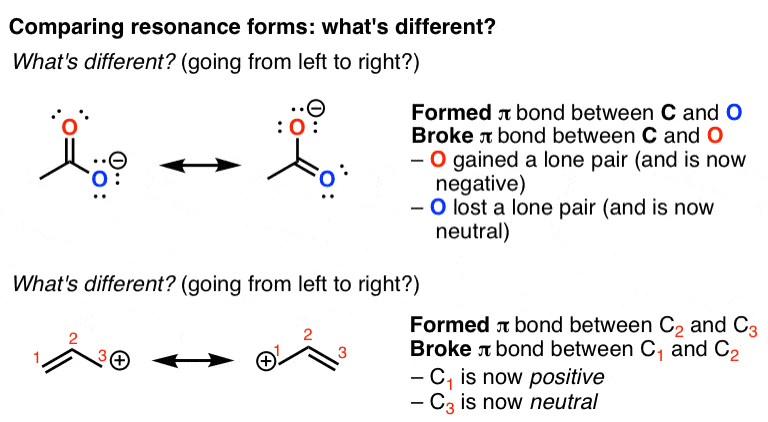
Most molecules and ions are represented by a single Lewis structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
